Chapter 14: Carbon element formation and chemistry. Also mystery of why Lithium isotope 6 is present in less quantity.
Electron distribution of Carbon = 1s2-2s2-2p2
Carbon is very important for all organism. Carbon have atomic number 6.
Carbon have following two isotopes stable.
Carbon 12= -C-12 = 6 neutrons +6 protons.
Carbon 13= C-13 = 7 Neutron + 6 Protons.
Abundance of C-12 is 98.0% and that of C-13 is 1.1%. Modern period table cannot explain the abundance distribution.
Carbon 12 isotope is formed by fusion of Lithium-6 isotope. And Carbon 12 isotope is formed by fusion of Lithium-6 and Lithium-7 isotopes.
|
Isotopes |
Lithium-6 |
Lithium-7 |
|
Lithium-6 |
Carbon-12 |
Carbon-13 |
|
Lithium-7 |
Carbon-13 |
Carbon-14 |
Abundance of Carbon-12: 2 Carbon-13: Carbon-14
Since neutrons is very high, fusion and formation of Carbon-13 and Carbon is difficult to form. Hence Carbon -12 abundance is very high and as Lithium 6 isotope are consumed, abundance of Lithium-6 is less.
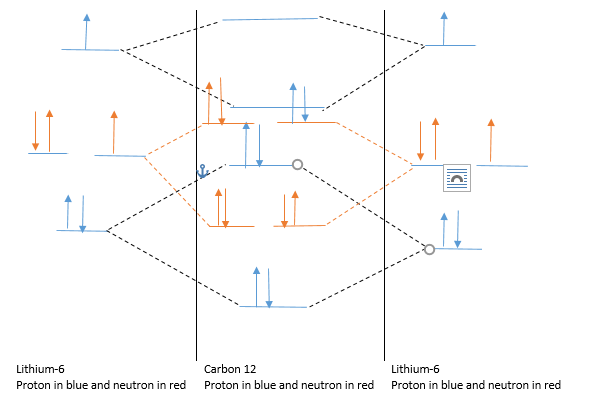
Carbon 12 formation :
Lithium-6 form bonding and anti bonding orbital with another Lithium-6 ion to give Carbon-12. Details are as follows.
Carbon 13 formation :
Lithium-7 form bonding and anti-bonding orbital with another Lithium-6 ion to give Carbon-12. Details are as follows.
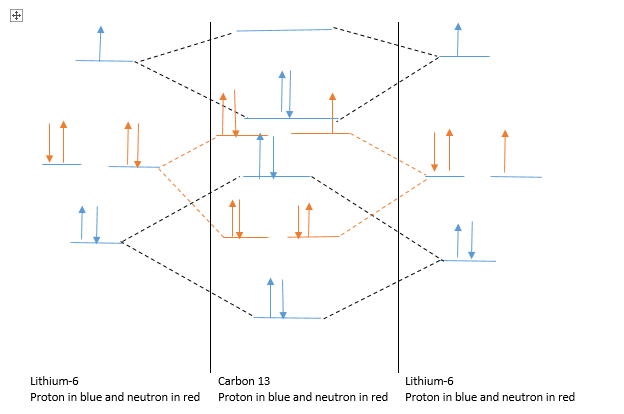
From the nuclear obital formation it is clear, C13 formation is difficult and hence, less formed. Also this explain why in nature abundance of Lithium-7 isotope is more compare to Lithium-6.
|
Natural effects |
Modern period table explanation |
Maneesh Dhauni Periodic table explanation |
|
Why abundance of Carbon 12 is greater than abundance of Carbon 13. |
No |
Yes. Explain very well. |
|
Explain why lithium 6 abundance is less than Lithium -7. |
No |
Yes Explain very well. |
|
Explain why Carbon have two paired proton and two paird electron at last orbital. Due to same energy level for last protons, I can have both proton to be unpaid. |
No |
Yes Explain very well. |